AI analyzes large volumes of lecture slides and scientific materials in minutes*1. With detection accuracy above 99%*2, it strongly supports human review and helps minimize the risk of missed safety signals.
Trial detection available
Contact Us
*1... Estimated processing time assumes standard file sizes. Larger volumes or more complex files may take longer.
*2... Accuracy may vary depending on product type, source material, and context. To reduce missed detections from a safety perspective, the AI may produce a certain level of over-detection.
It is becoming difficult to sustain a manual review operation that can reliably support deadline-based reporting.

Outsourcing to CROs creates recurring annual costs that can reach tens or even hundreds of millions of yen.

Reviewing large volumes of materials and extracting information requires significant time and labor, pushing teams close to their operational limit.

Many teams hesitate to adopt AI because of concerns about missed detections, false positives, fit with company rules, and final accountability.
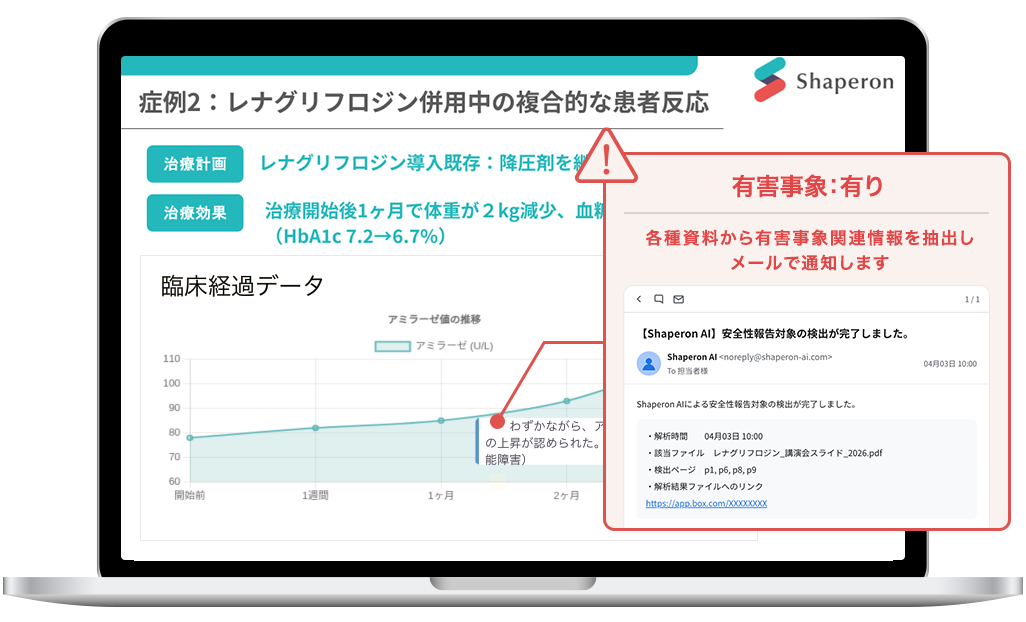
Simply upload a file and the AI will analyze whether adverse events are present in minutes*1, then notify you of the results.
Just upload files such as congress presentations and lecture slides. The service supports PDF, Word, PowerPoint, Excel, and other common formats.
It handles cross-page context and chronological interpretation, enabling high-accuracy detection even in complex document structures.
You receive an email summary of the decision results, and can access a linked report in PPT or Excel format showing the relevant passages and the reasons for each judgment.
*1... Estimated processing time assumes standard file sizes. Larger volumes or more complex files may take longer.
*2... Accuracy may vary depending on product type, source material, and context. To reduce missed detections from a safety perspective, the AI may produce a certain level of over-detection.
*2... Accuracy may vary depending on product type, source material, and context. To reduce missed detections from a safety perspective, the AI may produce a certain level of over-detection.
Consult us about any safety-information workflow challenge
AI analyzes materials before a human reviewer does. By identifying where adverse events are likely to appear across large volumes of material, it sharply reduces the time spent searching for relevant passages.

Use AI after human review to validate the result. The double-check helps prevent missed signals and also provides reassurance when the AI likewise finds no reportable event.

90% of pharmacovigilance
professionals
said the service detects adverse events without misses.
When the system also found nothing, we were able to conclude with confidence that no case was present.
Using it as a support tool shortened the time needed to confirm missing adverse events and identify relevant cases.
The AI detected adverse-event descriptions embedded in tables and graphs that had been missed during outsourced review.
*3... This content is based on feedback and survey responses from clients and has been edited for presentation.
In addition to further improving detection accuracy, we plan to gradually expand the service toward semi-automating the workflow from adverse-event detection through reporting-form entry.
Comprehensively extract adverse events, patient identifiers, outcomes, causality, and other details linked to suspect-drug cases to support entry into reporting formats.
Detect not only adverse events but also special situations such as pregnancy exposure, product-use errors, and drug interactions.
Detect safety information across diverse channels and formats, including daily reports, scientific papers, call centers, and chat logs.
Contact
Start by contacting us. We will discuss options based on your challenges and current operational workflow.
Free trial
Using your actual materials, you can confirm the AI’s accuracy and output format.
Initial setup
If the free trial meets your needs, we tune the model for implementation. We spend about one to three months adjusting it to your company-specific decision criteria and validating operations.
Go live
After launch, we continue improving accuracy to support your safety-information management workflow over time.
Trial detection available
Contact Us[Important notes]
This service is intended to support detection and does not replace pharmacovigilance operations. Final confirmation and judgment remain with human reviewers, and the accuracy and specifications may change over time.